A) acid catalysts
B) base catalysts
C) nucleophilic catalysts
D) metal ion catalysts
E) all of the above
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is an intermediate in the acid-catalyzed hydrolysis of an ester?
A) ![]()
B) ![]()
C) ![]()
D) Intermediates B and C
E) Intermediates A, B, and C
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which factor contributes to the catalytic activity of enzymes?
A) Reacting groups are brought together at the active site in the proper orientation.
B) Amino acid side chains serve as catalysts.
C) Metal ions in the active site act as catalysts.
D) Amino acid side chains stabilize transition states and intermediates.
E) all of the above
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about enzyme-catalyzed isomerization of glucose-6-phosphate to fructose-6-phosphate is not true?
A) The mechanism is totally different from the laboratory version of the enediol rearrangement.
B) The first step is acid- and base-catalyzed ring opening.
C) The second step is a base-catalyzed enediol rearrangement.
D) The third step is acid- and base-catalyzed conversion of the enediol to a ketone.
E) The fourth step is acid- and base-catalyzed ring closure.
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What type of intramolecular catalysis is occurring in the following reaction? 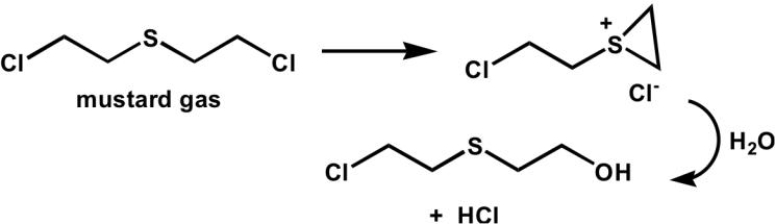
A) acid
B) base
C) nucleophilic
D) metal ion
E) none of the above
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about enzymatic catalysis is not true?
A) Enzymes are able to provide different solvent environments at the active site.
B) Enzymes allow one group to exist in its acidic form while a nearby group can exist in its basic form.
C) Solvent pH is unimportant in enzymatic reactions.
D) Enzyme activity depends on the reaction mixture pH.
E) A pH-rate profile plots enzyme activity versus pH.
G) A) and C)
Correct Answer

verified
C
Correct Answer
verified
Multiple Choice
Which of the following is not true about nucleophilic catalysis?
A) Nucleophilic catalysts form new covalent bonds with the reactants.
B) Nucleophilic catalysis will not involve positively charged intermediates.
C) Nucleophilic catalysis is also called covalent catalysis.
D) A nucleophilic catalyst increases the reaction rate by completely changing the mechanism of the reaction.
E) Nucleophilic catalysts are often good leaving groups.
G) All of the above
Correct Answer

verified
B
Correct Answer
verified
Multiple Choice
Which of the following is not true about metal-ion catalysis?
A) It can increase the rate of a reaction by removing a proton from the reactant.
B) It can make a reaction center more susceptible to receiving electrons.
C) It can make a leaving group a weaker base and therefore a better leaving group.
D) It can increase the rate of a hydrolysis reaction by increasing the nucleophilicity of water.
F) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements about trypsin, chymotrypsin, and elastase are true?
A) They are serine proteases.
B) They are endopeptidases.
C) Serine is part of both tetrahedral intermediates in the enzymatic mechanism.
D) Acid, base, and nucleophilic catalysis are utilized in the overall mechanism for these proteases.
E) All of the above are true.
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following factors is important in determining the rate of intramolecular reactions?
A) number of collisions per unit time
B) fraction of molecules with sufficient energy
C) fraction of molecules with proper orientation
D) favorable conformation for reaction
E) all of the above
G) A) and E)
Correct Answer

verified
E
Correct Answer
verified
Showing 1 - 10 of 10
Related Exams