A) low, high
B) high, high
C) low, low
D) high, low
F) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine if H2O is a suitable reagent to protonate the following compound. 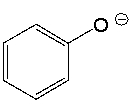
A) yes
B) no
D) undefined
Correct Answer

verified
Correct Answer
verified
Multiple Choice
As a result of the "leveling effect," the strongest acid that can exist in appreciable concentration in aqueous solution is ____.
A) H2SO4
B) HCl
C) H3O+
D) HNO3
E) All of these
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following solvents can be used with (CH3) 3CLi?
A) CH3CH2OH
B) CH3OH
C) H2O
D) Liquid NH3
E) CH3(CH2) 4CH3
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Provide a curved arrow mechanism for the following acid-base reaction. 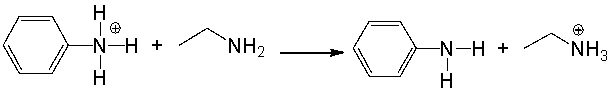
Correct Answer

verified
Correct Answer
verified
Essay
For the following reaction label the acid, base, conjugate acid and conjugate base. 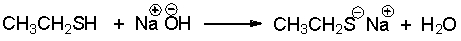
Correct Answer

verified
Correct Answer
verified
Essay
Draw the conjugate base of the following compound. 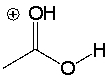
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following acid-base reaction, predict which side of the equilibrium is favored. 
A) favor right side
B) favor left side
C) neither
E) None of the above
Correct Answer

verified
Correct Answer
verified
Essay
Predict the products for the following acid-base reaction. 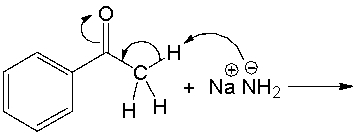
Correct Answer

verified
Correct Answer
verified
Essay
For the following acid-base reaction, predict which side of the equilibrium is favored. Explain why. 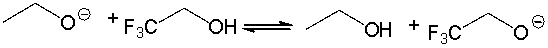
Correct Answer

verified
Favors the right side.
Both the base and...View Answer
Show Answer
Correct Answer
verified
Both the base and...
View Answer
Essay
For the following reaction, identify the Lewis acid and the Lewis base. 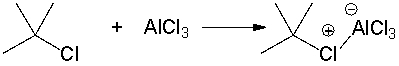
Correct Answer

verified
Correct Answer
verified
Essay
Which of the following compounds is more acidic? Explain why. CH3OH and CH3NH2
Correct Answer

verified
CH3OH is more acidic than CH3NH2. The conju...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which of the indicated protons is most acidic? 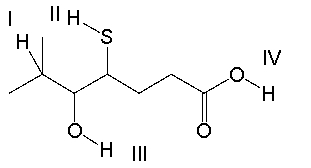
A) I
B) II
C) III
D) IV
E) All of these
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
For the following reaction, identify the Lewis acid. 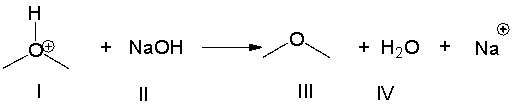
A) I
B) II
C) III
D) IV
E) none of these
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A loss of proton from a Brønsted-Lowry acid results in a ___.
A) Lewis acid
B) conjugate acid
C) conjugate base
D) conjugate acid-base pair
E) None of these
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the conjugate base in the following reaction. 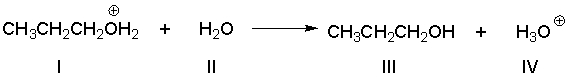
A) I
B) II
C) III
D) IV
E) none of the above
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
CH3CH2OCH2CH3, is best classified as a ____.
A) Brønsted-Lowry acid
B) Lewis acid
C) Brønsted-Lowry base
D) Lewis base
E) Both C & D
G) None of the above
Correct Answer

verified
Correct Answer
verified
Essay
Histidine an amino acid has the following structure. Circle the three most acidic protons in histidine. 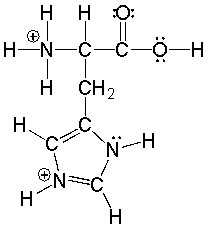
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following compounds is most basic? 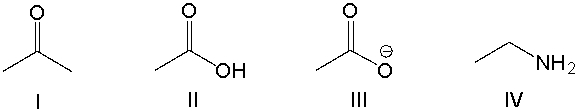
A) I
B) II
C) III
D) IV
E) None of these
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Essay
Aspartic acid, an amino acid, has the following structure. Circle the most acidic proton in aspartic acid. Explain your choice. 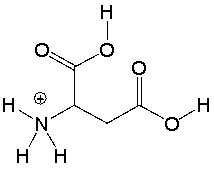
Correct Answer

verified
 _TB4454_00 The conju...
_TB4454_00 The conju...View Answer
Show Answer
Correct Answer
verified
View Answer
Showing 21 - 40 of 127
Related Exams