B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An iron ore sample is found to be 35.00% Fe by mass.How many grams of ore are needed to obtain 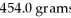 Of Fe?
Of Fe?
A) 1297
B) 158.9
C) 295.1
D) 350.0
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
An iron chloride compound contains 55.85 grams of iron and 106.5 grams of chlorine.What is the most likely empirical formula for this compound?
A) FeCl
B) FeCl2
C) Fe2Cl
D) FeCl3
E) Fe3Cl
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Determine the correct empirical formula of a compound containing 26.68% carbon,2.24% hydrogen,and 71.08% oxygen.
A) CHO
B) CHO2
C) C0.5H0.5O1
D) C2H2O4
E) none of the above
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A 7.96 gram sample of silver reacts with oxygen to form 8.55 gram of the metal oxide.What is the formula of the oxide?
A) AgO
B) AgO2
C) Ag2O
D) Ag3O
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
A compound has a molar mass of 180.15 g/mol.Given the following percent composition,calculate the molecular formula:
A) CH3O2
B) CH2O
C) C3H6O3
D) C6H12O6
E) none of the above
G) C) and D)
Correct Answer

verified
Correct Answer
verified
True/False
One mole of nitrogen gas contains (2)× (6.022 × 1023)nitrogen atoms.
B) False
Correct Answer

verified
Correct Answer
verified
True/False
The mass of 2.0 moles of H2O is greater than the mass of 1.0 mole of CO2.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles are there in 82.5 grams of iron?
A) 4.97 × 1025
B) 55.85
C) 0.677
D) 1.48
E) none of the above
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
If 3.011 × 1023 molecules have a mass of 20.04 grams,what is the molar mass of this substance?
A) 40.08 g/mol
B) 10.02 g/mol
C) 20.04 g/mol
D) 6.658 × 10-23 g/mol
E) none of the above
G) B) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Vitamin C is known chemically by the name ascorbic acid.Determine the empirical formula of ascorbic acid if it is composed of 40.92% carbon,4.58% hydrogen,and 54.50% oxygen.
A) CHO
B) CH2O
C) C2H3O2
D) C3H4O3
E) none of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the mass percent of oxygen in sodium oxide?
A) 17.0
B) 25.8
C) 44.4
D) 39.7
E) none of the above
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many atoms are in 1.50 moles of fluorine gas?
A) 6.022 × 1023
B) 9.03 × 1023
C) 18.98
D) 1.81 × 1024
E) none of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
The empirical formula mass is 18.0 and the molecular formula mass is 90,therefore n = 5.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles are there in 17.5 grams of sodium?
A) 22.99
B) 1.05 × 1025
C) 0.761
D) 1.31
E) none of the above
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many moles of iron are contained in 1.75 kg of iron?
A) 3.13 × 10-2
B) 3.13 × 10-4
C) 31.3
D) 3.13 × 104
E) none of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
True/False
Six grams of carbon contains 3.008 × 1023 atoms.
B) False
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the mass percent of hydrogen in water?
A) 33.3
B) 88.8
C) 5.60
D) 11.2
E) none of the above
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
What is the mass of 3.91 x 1024 sulfur atoms?
A) 208
B) 4.93
C) 155
D) 32.07
E) none of the above
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
You have 10.0 g each of Na,C,Pb,Cu and Ne.Which contains the smallest number of moles?
A) Na
B) C
C) Pb
D) Cu
E) Ne
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 111
Related Exams